2023-01-10
Realheart Reaches New Milestone With the World’s First Four Chamber Artificial Heart
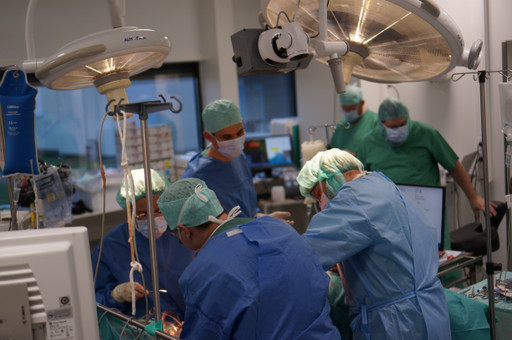
Scandinavian Real Heart AB, the company developing the world's first four-chamber total artificial heart (TAH), successfully extended the survival of animals implanted with Realheart’s TAH.
Since the series of animal trials with the clinical version of the company's TAH began in 2022, Realheart has been able to increase survival time from one to four days.
After implantation, the animal was able to stand up and eat. In addition, Scandinavian Real Heart was able to confirm key TAH performance criteria, including:
- No haemolysis (damage to red blood cells): Haemolysis due to mechanical stress caused by artificial hearts on blood cells is a major cause of severe side effects impacting survival of artificial heart patients over time.
- No thromboembolic events (blood clots): Good blood flow and low mechanical stress in the TAH minimise the risk of very serious, and life-threatening consequences for patients, including stroke.
- High pumping capacity and good right-left balance: The TAH can pump eight litres of blood per minute. This is significantly more capacity than what an adult human needs. The balance between blood flow from the right and left ventricles is crucial for survival.
- Short procedure: A heart-lung machine used during the implantation of the TAH was disconnected after less than 2.5 hours. The extended use of a heart-lung machine during open heart surgeries poses considerable safety risks to patients. Realheart is developing its TAH to reduce time on a heart-lung machine and overall procedure times, therefore improving overall safety.
''I am incredibly pleased with how much we have been able to achieve after just a few implantations of our TAH. The fact that we see no signs of haemolysis or blood clots, combined with the fact that we can achieve good left-right balance, a pulsatile blood flow, and natural blood pressure, is extremely satisfying. We are now moving forward with the aim of continuing to further prolong survival time by optimising the surgical methodology,'' said Ina Laura Perkins, CEO of Realheart. The company plans to initiate human clinical trials in 2024.
This disclosure contains information that Real Heart is obliged to make public pursuant to the EU Market Abuse Regulation (EU nr 596/2014). The information in this press release has been published through the agency of the contact persons set out below, at the time stated by Scandinavian Real Heart AB’s news distributor Cision upon publication of this press release.